For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics. Check with your local representative for availability in specific markets. 
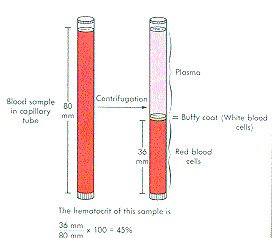
Not all products are available in all regions. Abstract METHODS We evaluated the analytical performance of the i-STAT through the measurement of sodi-um (Na), potassium (K), hematocrit, ionized calcium (iCa). Electrolyte, chemistry and hematocrit results are also depicted as bar graphs with reference ranges. IST LVL1 AQUS CTL(10X1.7) Product Number 06F1201. Item is Non - ReturnableLiquid QC and calibration verification, measures hematocrit, Blood gases, electrolytes, chemistries Item Id 810463 MF ID 05P7001. To share the full Product Catalog, please use the PRODUCT CATALOG > DOWNLOAD option from the navigation bar at the top of the page. GDPR Statement (last updated: May 2018) | Declaration for California Compliance Law. Istat Consumables Istat Consumables Use the PRINT, EMAIL and PDF buttons, to the right, to share only this page. Any person depicted in such photographs is a model. Photos displayed are for illustrative purposes only. With i-STAT CG8+, healthcare professionals can obtain blood gases, glucose, chemistries, hematocrit and hemoglobin levels in approximately two minutes without leaving the patient’s side. Causes of a Discrepancy Between a Hemoglobin or Hematocrit Value Determined on a Conductivity-Based Point-of-Care Instrument (iStat, Other) and Other. iSTAT is one of the frequently used POC devices to measure HCT and HBG in. Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy: US Citizens | Non-US Citizens. i-STAT CG8+ Cartridge Providing lab-quality results fast when every minute counts. Discrepancies in Hematocrit and Hemoglobin Measurements by PointofCare (POC). The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. A hematocrit test is part of a complete blood count (CBC). This website is governed by applicable U.S. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. Hemoglobin testing methods Hemocue method I-stat method Hematocrit General spun. Clinicians should not use the i-STAT(®) Hb in isolation for clinical decision-making when considering blood transfusion in a situation of 25% or greater blood loss.īlood loss haemoglobin measurement point-of-care.©2023 Abbott. Note: The i-STAT procedure is presented in Chapter 6, Chemistry. The standard deviation of i-STAT(®) Hb was greater after ≥25% estimated total blood volume loss. The i-STAT(®) Hb had an acceptable coefficient of variation, but the Hb levels were lower than those estimated by the laboratory. ISTAT TESTING PROCEDURE: 2 Athlete or Race Participant Testing Carefully insert cartridge into port of I-STAT analyzer. The measured conductivity, after correction for electrolyte concentration, is inversely related to the hematocrit. There was poor correlation between total plasma protein and bias in i-STAT(®) measurements. Hematocrit is determined conductometrically. Abbotts i-STAT was the most common POCT device used for blood gas. Primary Container: Lithium Heparin PST Gel (Light Green) or Lithium Heparin. oxygen saturation and hematocrit and requires a sample size of just 68l. If tests are repeated within 5 minutes, only the second set of results are allowed to stay as the permanent record. Test Name/Synonym: Hematocrit (HCT) iSTAT, Blood - North Zone. These errors are transmitted to EPIC system. The mean total plasma protein difference (total plasma protein T=0 minus T=1) was 13.6 g/l (95% confidence interval 10.2 to 17.0). Results with, >, < or <> - if persistent do not use ISTAT for testing.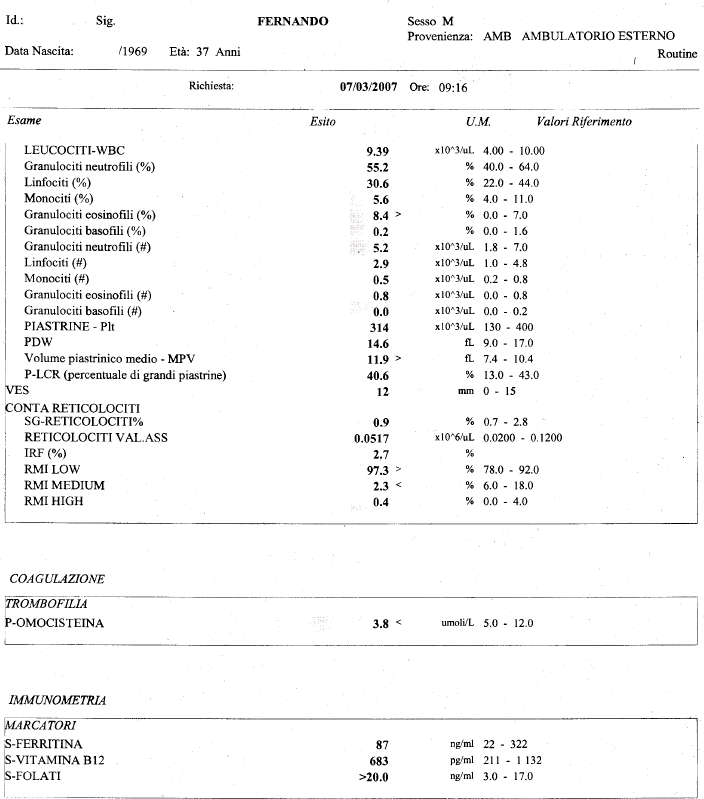
The mean difference between i-STAT(®) and laboratory Hb was -7.6 g/l (standard deviation 6.5) at T=0 and -5.1 g/l (standard deviation 12) at T=1. The coefficient of variation of the paired i-STAT(®) Hb estimates was 2.8% and 2.9% at T=0 and T=1, respectively. Blood tests for i-STAT(®) Hb, laboratory Hb (Sysmex XE-2100(™), Sysmex Corporation, Kobe, Japan) and total plasma proteins were obtained at the start of surgery (T=0) and when an estimated 25% total blood volume loss had occurred (T=1). We investigated the accuracy of i-STAT(®) (Abbott Point of Care Inc., Princeton, NJ, USA) haemoglobin (Hb) measurement in surgical patients with an estimated blood loss of ≥25% of total blood volume.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
